CAR (chimeric antigen receptor) T-cell therapy aims to re-engineer T-cells from your immune system so they can better target cancer.
We plan to offer CAR T-cell therapy at Epworth through haematology clinical trials later in 2023.
What is CAR T-cell therapy?
T cells are a type of white blood cell. They’re a part of your immune system and help your body fight infection. Their role is to recognise and destroy anything that shouldn’t be there (abnormal cells). Cancer cells are an abnormal cell.
But T-cells sometimes fail to recognise or effectively destroy cancer cells.
Scientists can genetically modify (change) your T-cells to make CAR T-cells for blood cancer treatment.
What is CAR T-cell therapy?
How it works
CAR T-cell therapy is not suitable for everyone. Your doctor will discuss the risks and help you decide if it’s right for you.
Step 1. Apheresis (cell collection)
CAR T-cell therapy uses T-cells collected from your body using apheresis.
Step 2. CAR T-cell development – in the lab
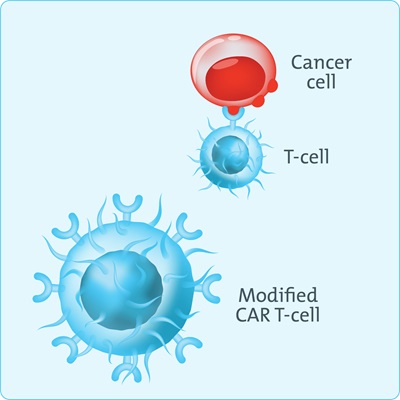
Scientists genetically engineer the T-cells to have chimeric antigen receptors (CAR) on their surface. The T-cells can now recognise a specific protein (antigen) on other cell surfaces.
Example
The CD19 antigen is found in most B-cell lymphomas. A CAR T-cell will look for all cells with the CD19 antigen and destroy them.
The CAR T-cell has the same job as your original T cell. But with the therapy, it can now do its job better.
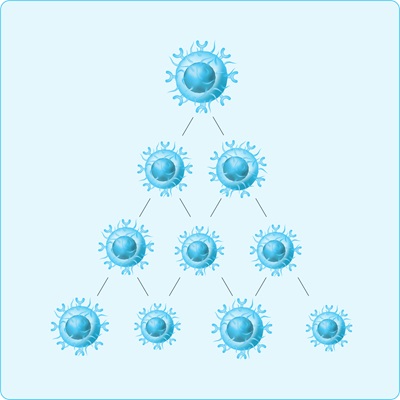
Step 3. Increase the number of CAR T-cells – in the lab
Scientists multiply the CAR T-cells so there’s millions to help recognise and destroy cancer cells later.
Step 4. Prepare for the therapy at Epworth
To receive the CAR T-cells, your body needs to have less T-cells than normal. A short course of chemotherapy prepares your body so you’re ready for the therapy.
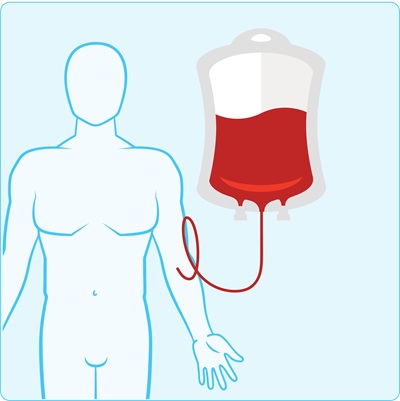
Step 5. Receive CAR T-cell therapy
The CAR T-cells are returned into your body through an infusion. Once they’re in your body, they continue to multiply and look for cancer cells to destroy.
CAR T-cell therapy has successfully put some blood cancer patients into remission, even when other treatments have stopped working.
For some people, CAR T-cells stay in the body and are ready to destroy cancer cells if they come back.
CAR T-cell approvals and funding in Australia
CAR T-cell therapy is currently approved and publicly funded in Australia for certain relapsed or refractory B-cell lymphomas:
- Diffuse large B-cell lymphoma in adults
- Transformed follicular lymphoma in adults
- Primary mediastinal B-cell lymphoma in adults
- B-cell acute lymphoblastic leukaemia for children and young adults up to the age of 25
Clinical trials continue to research CAR T-cell therapy as a treatment for other types of blood cancers and some solid tumours.